Why are audits and CAPA follow-up essential for Biotech companies?
Why are audits and CAPA follow-up essential for Biotech companies?
Why are audits and CAPA follow-up essential for Biotech companies?
Wednesday, March 18, 2026
Wednesday, March 18, 2026

Why are audits and CAPA follow-up essential for Biotech companies?
Key elements for compliance, risk management, and operational excellence
In the biotechnology and pharmaceutical industries, sponsors face a regulatory imperative: they must ensure full control over their subcontractors. Audits are not just a compliance checkbox — they are a strategic tool to safeguard quality, mitigate risks, and drive continuous improvement.
At INITS, we deliver more than audits. Our comprehensive, customized auditing services are backed by rigorous CAPA follow-up — a critical differentiator that adds tangible value to your supplier monitoring.
We follow a meticulous audit process designed to ensure clarity, compliance, and continuity
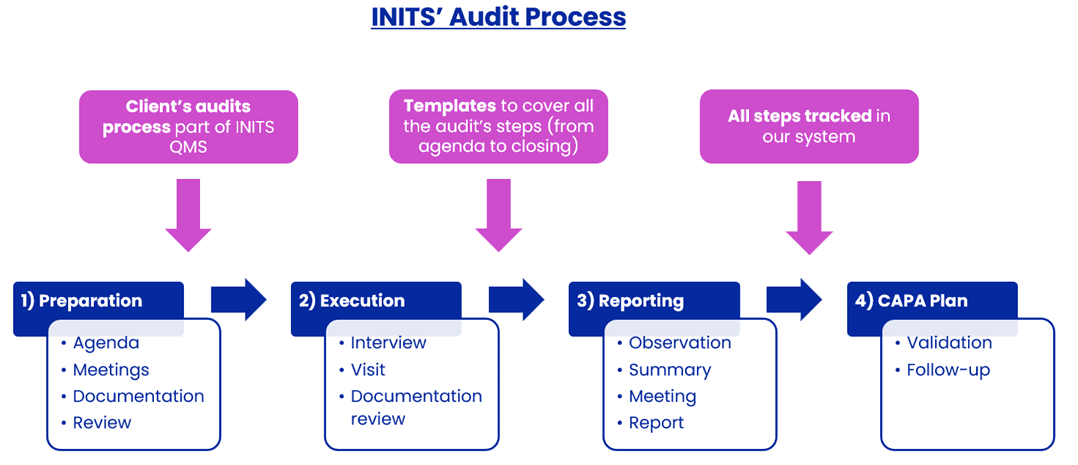
Our proven methodology, refined through over 300 audits, covers all critical activities:
Scope of activities: Storage, quality control, manufacturing, packaging, release, logistics, and more.
Product types: Cell banks, drug substances, drug products, bio/non-bio, sterile/non-sterile, investigational medicinal products, etc.
Audit modalities: On-site (international) and remote audits, tailored to your needs
Our team — comprising qualified auditors, quality officers, and audit referent — ensures deep expertise and a results-driven approach, fully aligned with GxP, ICH, FDA, and EMA standards.
CAPA follow-up: A real differentiator for compliance and efficiency
At INITS, we provide end-to-end CAPA management, helpings clients:
Save internal resources by reducing administrative burdens.
Enhance compliance and inspection readiness with full traceability for inspections.
Keep auditees accountable with proactive follow-up.
This service is especially valued by organizations with limited in-house QA capacity or those managing multiple audits simultaneously.
Let’s talk!
Discover how our audits and CAPA services can simplify your quality processes and free up your team.
#INITS #PharmaAudit #CAPAFollowUp #PharmaceuticalCompliance #GMP #QualityAssurance #RegulatoryAffairs #Biomanufacturing #PharmaServices #AuditingExcellence
Why are audits and CAPA follow-up essential for Biotech companies?
Key elements for compliance, risk management, and operational excellence
In the biotechnology and pharmaceutical industries, sponsors face a regulatory imperative: they must ensure full control over their subcontractors. Audits are not just a compliance checkbox — they are a strategic tool to safeguard quality, mitigate risks, and drive continuous improvement.
At INITS, we deliver more than audits. Our comprehensive, customized auditing services are backed by rigorous CAPA follow-up — a critical differentiator that adds tangible value to your supplier monitoring.
We follow a meticulous audit process designed to ensure clarity, compliance, and continuity

Our proven methodology, refined through over 300 audits, covers all critical activities:
Scope of activities: Storage, quality control, manufacturing, packaging, release, logistics, and more.
Product types: Cell banks, drug substances, drug products, bio/non-bio, sterile/non-sterile, investigational medicinal products, etc.
Audit modalities: On-site (international) and remote audits, tailored to your needs
Our team — comprising qualified auditors, quality officers, and audit referent — ensures deep expertise and a results-driven approach, fully aligned with GxP, ICH, FDA, and EMA standards.
CAPA follow-up: A real differentiator for compliance and efficiency
At INITS, we provide end-to-end CAPA management, helpings clients:
Save internal resources by reducing administrative burdens.
Enhance compliance and inspection readiness with full traceability for inspections.
Keep auditees accountable with proactive follow-up.
This service is especially valued by organizations with limited in-house QA capacity or those managing multiple audits simultaneously.
Let’s talk!
Discover how our audits and CAPA services can simplify your quality processes and free up your team.
#INITS #PharmaAudit #CAPAFollowUp #PharmaceuticalCompliance #GMP #QualityAssurance #RegulatoryAffairs #Biomanufacturing #PharmaServices #AuditingExcellence
Why are audits and CAPA follow-up essential for Biotech companies?
Key elements for compliance, risk management, and operational excellence
In the biotechnology and pharmaceutical industries, sponsors face a regulatory imperative: they must ensure full control over their subcontractors. Audits are not just a compliance checkbox — they are a strategic tool to safeguard quality, mitigate risks, and drive continuous improvement.
At INITS, we deliver more than audits. Our comprehensive, customized auditing services are backed by rigorous CAPA follow-up — a critical differentiator that adds tangible value to your supplier monitoring.
We follow a meticulous audit process designed to ensure clarity, compliance, and continuity

Our proven methodology, refined through over 300 audits, covers all critical activities:
Scope of activities: Storage, quality control, manufacturing, packaging, release, logistics, and more.
Product types: Cell banks, drug substances, drug products, bio/non-bio, sterile/non-sterile, investigational medicinal products, etc.
Audit modalities: On-site (international) and remote audits, tailored to your needs
Our team — comprising qualified auditors, quality officers, and audit referent — ensures deep expertise and a results-driven approach, fully aligned with GxP, ICH, FDA, and EMA standards.
CAPA follow-up: A real differentiator for compliance and efficiency
At INITS, we provide end-to-end CAPA management, helpings clients:
Save internal resources by reducing administrative burdens.
Enhance compliance and inspection readiness with full traceability for inspections.
Keep auditees accountable with proactive follow-up.
This service is especially valued by organizations with limited in-house QA capacity or those managing multiple audits simultaneously.
Let’s talk!
Discover how our audits and CAPA services can simplify your quality processes and free up your team.
#INITS #PharmaAudit #CAPAFollowUp #PharmaceuticalCompliance #GMP #QualityAssurance #RegulatoryAffairs #Biomanufacturing #PharmaServices #AuditingExcellence
© 2024 INITS. All rights reserved
© 2024 INITS. All rights reserved
© 2024 INITS. All rights reserved
