Reference Standards: The Cornerstone of Quality and Compliance in Pharmaceutical Analysis
Reference Standards: The Cornerstone of Quality and Compliance in Pharmaceutical Analysis
Reference Standards: The Cornerstone of Quality and Compliance in Pharmaceutical Analysis
Tuesday, April 28, 2026
Tuesday, April 28, 2026

Reference Standards: The Cornerstone of Quality and Compliance in Pharmaceutical Analysis
A Reference Standard is far more than just a calibrant — it is the foundation of reliable quantitative testing, identity verification, and purity assessment. Its quality must be fit for purpose, ensuring robust manufacturing control and traceability. Unlike routine testing, Reference Standards undergo additional, rigorous characterization to fulfill their critical role.
Their use isn’t optional - it is a regulatory imperative. Both IND and IMPD submissions require a dedicated Reference Standard section. According to ICH Q6B and ICH Q7, it is unlikely that an international or national standard will fully represent a new molecular entity. Manufacturers must therefore establish an in-house primary reference material (derived from representative production and clinical lots) by the time of submission.
Strategic Management Across the Product Lifecycle
Managing Reference Standards is a critical and dynamic process. Multiple reference materials may be implemented for various reasons:
Small-scale batch manufacturing, limiting material availability.
Batch aging, which can compromise integrity.
Process changes, potentially affecting representativeness.
A clear, proactive strategy is essential to ensure data consistency throughout the product’s life.
Implementation and Monitoring: A Rigorous Approach
1. A tailored implementation by Clinical Stage
Requirements evolve with clinical progress, depending on CQAs and product knowledge as shown in the Figure below:
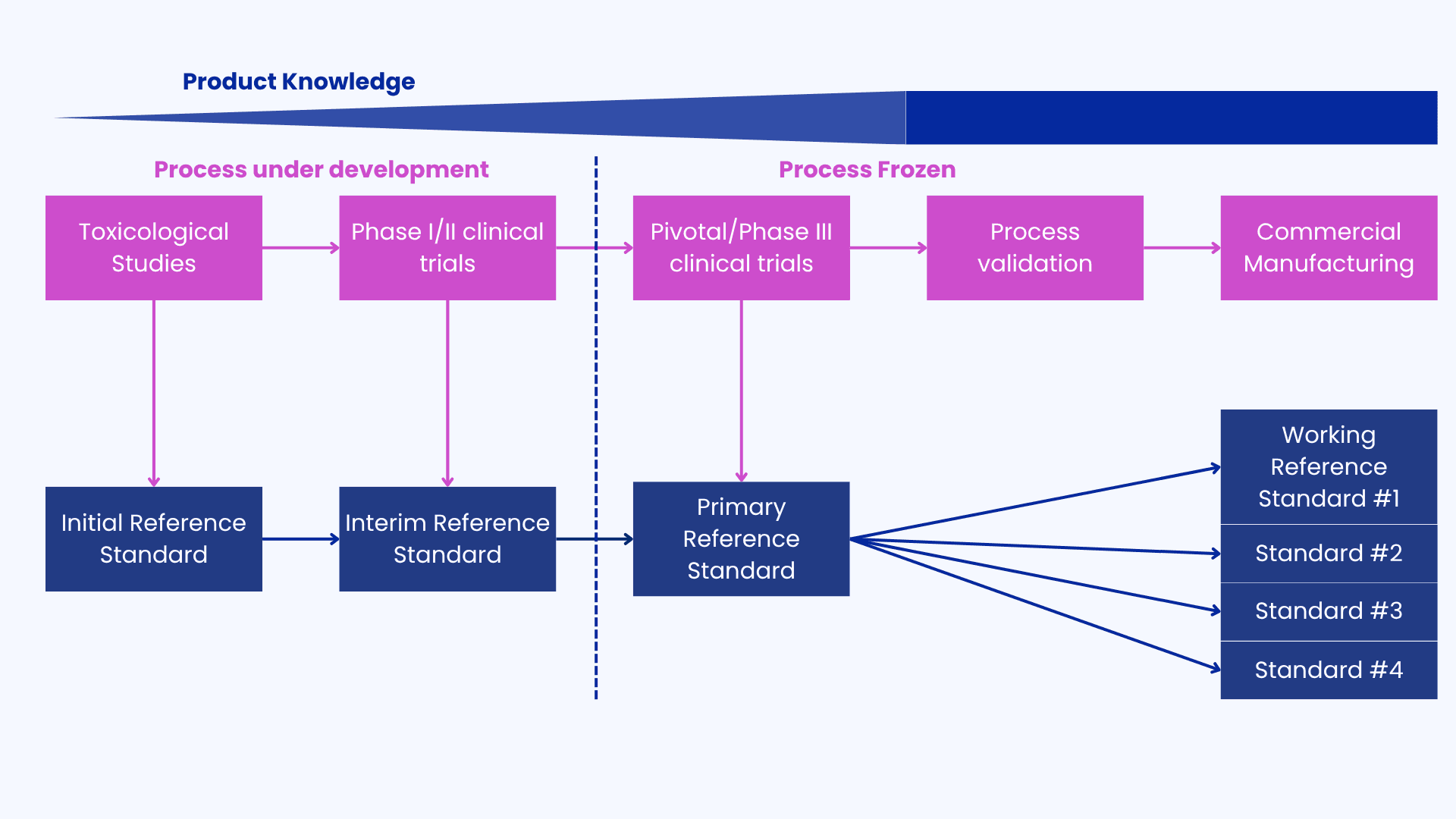
2. Ongoing Monitoring
Vigilant oversight is crucial to maintain key functions (e.g., titer, biological activity) and prevent drift. It can be performed through control card establishment.
3. Bridging Between Standards: Ensuring Comparability
Transitioning from one standard to another demands a well-defined protocol, including:
Batch selection: must be representative of the current process, free from deviations that may affect its quality, and sufficient in quantity for release and stability testing of subsequent batches.
Assays to perform: need to cover all relevant CQA parameters.
Comparison method: should be side-by-side or CoA-based.
Number of assays: must be determined by method variability and parameter significance.
Acceptance criteria: must be based on specifications, possibly supplemented by tolerance intervals derived from the current Reference Standard.
These activities are critical and must be anticipated, as implementing a new Reference Standard involves both manufacturing and control of a dedicated batch.
4. Two-Tiered System: The best practice
ICH guidelines recommend a two-tiered system to secure the bridging of Reference Standards. This approach establishes a primary standard (the gold standard) against which all working standards are calibrated. The primary standard, representative of the commercial process, undergoes extensive additional characterization beyond routine batch release testing.
5. QMS: a critical tool for Ref Mat management
The Quality Management System defines, justifies, and drives strategy while ensuring its effective implementation. Through change control, it manages the comparability, implementation, and supply of reference standards; risk assessment oversees shelf-life and deviation management and SOPs and forms ensure consistent process application.
Management of Reference Standards is a multi-disciplinary exercise involving CMC, Regulatory, Quality and Supply – expertise integrated across our dedicated teams at INITS.
Want to learn more about reference standard or explore how INITS can support your Reference Standard management? Feel free to reach out to us!
#RegulatoryCompliance #Biotech #CMC #GMP #ReferenceStandards #DrugDevelopment
Reference Standards: The Cornerstone of Quality and Compliance in Pharmaceutical Analysis
A Reference Standard is far more than just a calibrant — it is the foundation of reliable quantitative testing, identity verification, and purity assessment. Its quality must be fit for purpose, ensuring robust manufacturing control and traceability. Unlike routine testing, Reference Standards undergo additional, rigorous characterization to fulfill their critical role.
Their use isn’t optional - it is a regulatory imperative. Both IND and IMPD submissions require a dedicated Reference Standard section. According to ICH Q6B and ICH Q7, it is unlikely that an international or national standard will fully represent a new molecular entity. Manufacturers must therefore establish an in-house primary reference material (derived from representative production and clinical lots) by the time of submission.
Strategic Management Across the Product Lifecycle
Managing Reference Standards is a critical and dynamic process. Multiple reference materials may be implemented for various reasons:
Small-scale batch manufacturing, limiting material availability.
Batch aging, which can compromise integrity.
Process changes, potentially affecting representativeness.
A clear, proactive strategy is essential to ensure data consistency throughout the product’s life.
Implementation and Monitoring: A Rigorous Approach
1. A tailored implementation by Clinical Stage
Requirements evolve with clinical progress, depending on CQAs and product knowledge as shown in the Figure below:

2. Ongoing Monitoring
Vigilant oversight is crucial to maintain key functions (e.g., titer, biological activity) and prevent drift. It can be performed through control card establishment.
3. Bridging Between Standards: Ensuring Comparability
Transitioning from one standard to another demands a well-defined protocol, including:
Batch selection: must be representative of the current process, free from deviations that may affect its quality, and sufficient in quantity for release and stability testing of subsequent batches.
Assays to perform: need to cover all relevant CQA parameters.
Comparison method: should be side-by-side or CoA-based.
Number of assays: must be determined by method variability and parameter significance.
Acceptance criteria: must be based on specifications, possibly supplemented by tolerance intervals derived from the current Reference Standard.
These activities are critical and must be anticipated, as implementing a new Reference Standard involves both manufacturing and control of a dedicated batch.
4. Two-Tiered System: The best practice
ICH guidelines recommend a two-tiered system to secure the bridging of Reference Standards. This approach establishes a primary standard (the gold standard) against which all working standards are calibrated. The primary standard, representative of the commercial process, undergoes extensive additional characterization beyond routine batch release testing.
5. QMS: a critical tool for Ref Mat management
The Quality Management System defines, justifies, and drives strategy while ensuring its effective implementation. Through change control, it manages the comparability, implementation, and supply of reference standards; risk assessment oversees shelf-life and deviation management and SOPs and forms ensure consistent process application.
Management of Reference Standards is a multi-disciplinary exercise involving CMC, Regulatory, Quality and Supply – expertise integrated across our dedicated teams at INITS.
Want to learn more about reference standard or explore how INITS can support your Reference Standard management? Feel free to reach out to us!
#RegulatoryCompliance #Biotech #CMC #GMP #ReferenceStandards #DrugDevelopment
Reference Standards: The Cornerstone of Quality and Compliance in Pharmaceutical Analysis
A Reference Standard is far more than just a calibrant — it is the foundation of reliable quantitative testing, identity verification, and purity assessment. Its quality must be fit for purpose, ensuring robust manufacturing control and traceability. Unlike routine testing, Reference Standards undergo additional, rigorous characterization to fulfill their critical role.
Their use isn’t optional - it is a regulatory imperative. Both IND and IMPD submissions require a dedicated Reference Standard section. According to ICH Q6B and ICH Q7, it is unlikely that an international or national standard will fully represent a new molecular entity. Manufacturers must therefore establish an in-house primary reference material (derived from representative production and clinical lots) by the time of submission.
Strategic Management Across the Product Lifecycle
Managing Reference Standards is a critical and dynamic process. Multiple reference materials may be implemented for various reasons:
Small-scale batch manufacturing, limiting material availability.
Batch aging, which can compromise integrity.
Process changes, potentially affecting representativeness.
A clear, proactive strategy is essential to ensure data consistency throughout the product’s life.
Implementation and Monitoring: A Rigorous Approach
1. A tailored implementation by Clinical Stage
Requirements evolve with clinical progress, depending on CQAs and product knowledge as shown in the Figure below:

2. Ongoing Monitoring
Vigilant oversight is crucial to maintain key functions (e.g., titer, biological activity) and prevent drift. It can be performed through control card establishment.
3. Bridging Between Standards: Ensuring Comparability
Transitioning from one standard to another demands a well-defined protocol, including:
Batch selection: must be representative of the current process, free from deviations that may affect its quality, and sufficient in quantity for release and stability testing of subsequent batches.
Assays to perform: need to cover all relevant CQA parameters.
Comparison method: should be side-by-side or CoA-based.
Number of assays: must be determined by method variability and parameter significance.
Acceptance criteria: must be based on specifications, possibly supplemented by tolerance intervals derived from the current Reference Standard.
These activities are critical and must be anticipated, as implementing a new Reference Standard involves both manufacturing and control of a dedicated batch.
4. Two-Tiered System: The best practice
ICH guidelines recommend a two-tiered system to secure the bridging of Reference Standards. This approach establishes a primary standard (the gold standard) against which all working standards are calibrated. The primary standard, representative of the commercial process, undergoes extensive additional characterization beyond routine batch release testing.
5. QMS: a critical tool for Ref Mat management
The Quality Management System defines, justifies, and drives strategy while ensuring its effective implementation. Through change control, it manages the comparability, implementation, and supply of reference standards; risk assessment oversees shelf-life and deviation management and SOPs and forms ensure consistent process application.
Management of Reference Standards is a multi-disciplinary exercise involving CMC, Regulatory, Quality and Supply – expertise integrated across our dedicated teams at INITS.
Want to learn more about reference standard or explore how INITS can support your Reference Standard management? Feel free to reach out to us!
#RegulatoryCompliance #Biotech #CMC #GMP #ReferenceStandards #DrugDevelopment
© 2024 INITS. All rights reserved
© 2024 INITS. All rights reserved
© 2024 INITS. All rights reserved
